Dean Ho, director of Singapore’s N.1 Institute for Health, has used machine learning to discover the optimal combination of drugs to treat the coronavirus. Here’s how he did it
The words hydroxychloroquine and azithromycin have been splashed across almost every major news site over the last week, following US president Donald Trump’s announcement that he is taking both drugs to ward off the coronavirus—despite there being no evidence that the drugs are effective.
Hydroxychloroquine treats both malaria and arthritis, while azithromycin is an antibiotic, intended to prevent infections. Though both drugs have been approved for human use, neither have been proven as a viable treatment for the coronavirus.
Public interest in the drugs spiked soon after Trump’s televised address, sparking health officials and scientists to warn that the drugs may be unsafe and shouldn’t be taken at home, emphasising that trials for hydroxychloroquine showed potential fatal heart complications.
Dean Ho, the director of N.1 Institute for Health, an infectious diseases programme within the National University of Singapore, agrees that both drugs are relatively ineffective in the fight against the coronavirus. “Data suggests that hydroxychloroquine may be linked to cardiac issues and at this time, it hasn’t been shown to improve outcomes for patients,” says Ho, who is also head of the university's department of Biomedical Engineering.
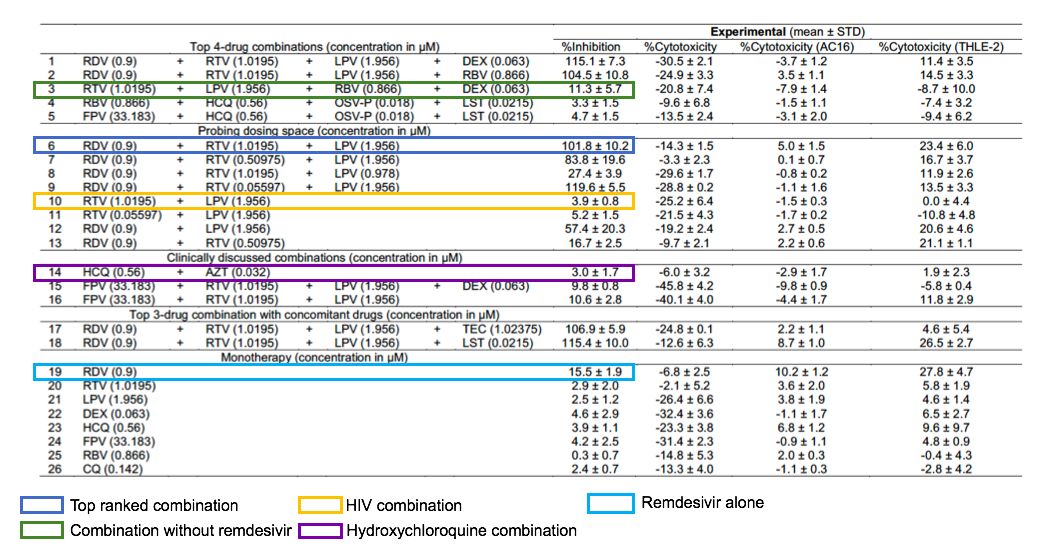
Instead, Ho believes the optimal treatment for the coronavirus is a combination of three drugs: lopinavir and ritonavir, which are currently used to treat HIV, and remdesivir, an antiviral drug originally used to treat ebola.

Ho and his multidisciplinary team at the N.1 Institute for Health determined the optimal coronavirus treatment using their artificial intelligence platform IDentif.AI, which uses machine learning to identify optimal infectious disease therapies. In a matter of days, IDentif.AI was able to test and identify trillions of different drug doses and combinations, providing clinicians with a list of drug treatments, ranging from most optimal to least optimal. It’s important to provide clinicians with a range of drug treatments, says Ho, as it allows them to counter any potential drug supply shortages, financial restrictions or patient allergies.
The AI testing was carried out on a live strain of the coronavirus, taken from a patient in Singapore, and was studied alongside 12 clinically actionable drugs on a kidney cell—a cell type often used to study viral infections. IDentif.AI found that the combination of lopinavir, ritonavir and remdesivir had an inhibition percentage of virtually 100 percent on average—indicating that the coronavirus was completely neutralised, while the combination of hydroxychloroquine and azithromycin was relatively ineffective, at just 3 percent inhibition.